23 developed a block-based fast warping tool that successfully aligned several GB-volume datasets. Thus, the strategy of obtaining the transformation parameters at low resolution and warping massive amounts of data in blocks is a better choice.
Thebrain 9 import outline registration#
Clearly, the nonlinear registration algorithm is a global optimization solution that consumes large amounts of memory 22. For example, in one study 21 registering a 12.5 μm 3 resolution whole-brain dataset approximate to 1 GB. Generally, the registration tools such as ITK that are widely used in biomedical fields are suitable for the registration of GB-scale data volumes. Briefly, when neuroscientists are struggling to obtain a valuable experimental dataset, they often find that it is difficult to extract sufficient feature points accurately to ensure the registration quality.Īnother challenge occurs with the TB-scale large-volume whole-brain datasets brought by imaging at the cellular level. 17 combined the benefits of artificial and automatic methods and achieved the registration of two-dimensional continuous microscopy images to a brain atlas. 20 used automatic methods to extract feature points and achieved the registration of histopathological data, but the accuracy of the automatic recognition methods used was greatly affected by image quality. 14 completed the registration of two-dimensional microscopy images to MRI data by manually extracting feature points, but their number was limited and subjective. Hence, for the more complex optical microscopy images, feature-based registration algorithms 17, 18, 19 are the better choice because the factors that affect registration accuracy can be changed from the grayscale level to the extracted features level then, the bottleneck involves accurately and objectively extracting sufficient features. Similarly optical microscopy images are susceptible to variations in sample preparation and imaging processes. However, the gray-level-based methods are highly dependent on the quality of the original image signals.
Thebrain 9 import outline serial#
16 processed serial two-photon (STP) datasets to obtain an average brain. These methods can also be applied to optical microscopic images. Among these, gray-level-based registration algorithms 11 can effectively achieve nonlinear registration of MRI datasets with uniform signals. Previous studies 11, 12, 13, 14, 15 have been conducted to solve the registration of whole-brain three-dimensional datasets, especially within the MRI field. In addition, given the large-volume datasets produced during imaging, we urgently need a robust nonlinear registration pipeline that can register massive spatial information datasets with cellular resolution. However, with the rapid development of high-resolution whole-brain microscopic imaging 7, 8, 9, the obvious heterogeneous and nonuniform 10 characteristics of brain structures at the cellular level make it difficult to map the variety of experimental datasets from different individuals and modalities to a standard brain space using the uniform registration methods 11 that have been used effectively in previous macroscopic datasets, such as magnetic resonance imaging (MRI) datasets. Drawing a stereotaxic brain atlas 5, 6 provides a unified spatial reference for addressing this issue. Creating a comprehensive space in which various brains are mapped encompasses complex spatiotemporal information that can greatly facilitate the comparison 2, reuse 3 and integration 4 of brain datasets. Mapping brain structures in three dimensions is necessary to thoroughly understand brain functions 1. BrainsMapi will facilitate the comparison, reuse and integration of a variety of brain datasets.

In addition, it can register and integrate unregistered vectorized datasets into a common brain space.

By performing validation on model data and biological images, BrainsMapi achieves accurate registration on intramodal, individual, and multimodality datasets and can also complete the registration of large-volume datasets (approximately 20 TB) within 1 day. In this study, we propose a robust Brain Spatial Mapping Interface (BrainsMapi) to address the registration of large-volume datasets by introducing extracted anatomically invariant regional features and a large-volume data transformation method. However, because of the heterogeneous and nonuniform brain structure characteristics at the cellular level introduced by recently developed high-resolution whole-brain microscopy techniques, it is difficult to apply a single standard to robust registration of various large-volume datasets. Using the brain atlas as a hub, mapping detected datasets into a standard brain space enables efficient use of various datasets.
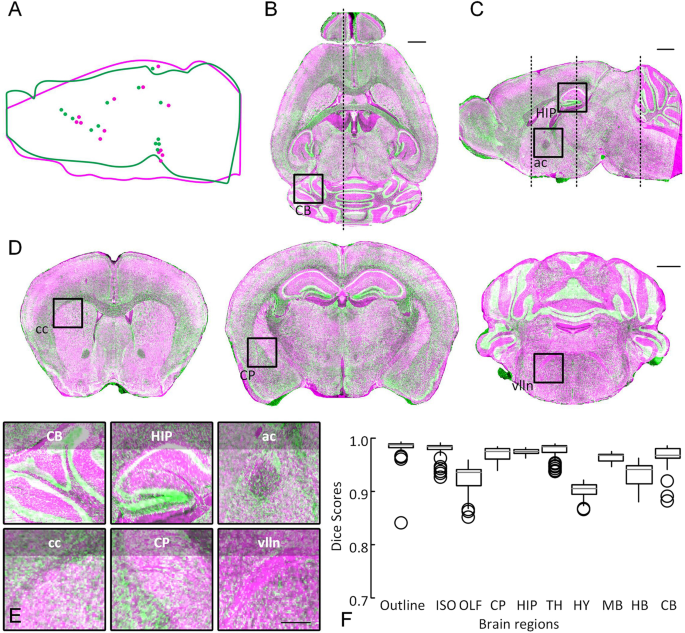
Accurately mapping brain structures in three-dimensions is critical for an in-depth understanding of brain functions.
